 
Point zero seven nine, and we get negative two point five four, so negative two point five four. Point zero seven nine, so the natural log of Log of all of these numbers, and before we graph something, right? So we need to take the natural log of point zero nine nine, so we get out the calculator here, so natural log of point zero nine nine gives us negative two point three one, so we put negative two Of cyclopropane, right? So right now we have only theĬoncentration of cyclopropane, we need to take the natural Graph that, all right, we need to figure out the natural log of the concentration Reaction is first-order, and the slope of that line, right, M, should be equal to negative K where K is your rate constant, and the Y intercept, right, should be equal to the natural log of the initial concentration In the previous video follows the form Y is equal to M X plus B, so if we put the natural log of the concentration ofĬyclopropane on the Y axis, and we put time on the X axis, if we do that, and we get a straight line, or close to a straight line, then we know that the Initial concentration of A, and for this reaction A is cyclopropane, so we could write this as the natural log of the concentration ofĬycolopropane, C three H six, is equal to negative K T plus the natural log of the initial concentration of cyclopropane, which we also talked about This is first-order, we need to use the integrated rate law from the previous video, so in the previous video we showed that the natural log of the concentration of A is equal to negative K T plus the natural log of the Show that it's first-order, so we look at the data over here, and we can see as time increases, right, the concentration ofĬyclopropane decreases, which makes sense because cyclopropane is turning into propene. The concentration of H 2O 2 decreases by half during each successive period of 6.00 hours.- Let's see how to plot data for a first-order reaction, so the conversion ofĬyclopropane into propene is a first-order reaction, and in part A they want us to use the experimental data to During the second half-life (from 6.00 hours to 12.00 hours), it decreases from 0.500 M to 0.250 M during the third half-life, it decreases from 0.250 M to 0.125 M. Using the decomposition of hydrogen peroxide in Figure 1 as an example, we find that during the first half-life (from 0.00 hours to 6.00 hours), the concentration of H 2O 2 decreases from 1.000 M to 0.500 M. In each succeeding half-life, half of the remaining concentration of the reactant is consumed. The half-life of a reaction ( t 1/2) is the time required for one-half of a given amount of reactant to be consumed. The correct time is 35 minutes! The Half-Life of a Reaction Aassuming no change in this zero-order behavior, you should be able to calculate the time (min) when the concentration will reach 0.0001 mol L −1. The zero-order plot in Figure 4 shows an initial ammonia concentration of 0.0028 mol L −1 decreasing linearly with time for 1000 s. Integration of the rate law for a simple first-order reaction (rate = k) results in an equation describing how the reactant concentration varies with time: For purposes of discussion, we will focus on the resulting integrated rate laws for first-, second-, and zero-order reactions. This process can either be very straightforward or very complex, depending on the complexity of the differential rate law. 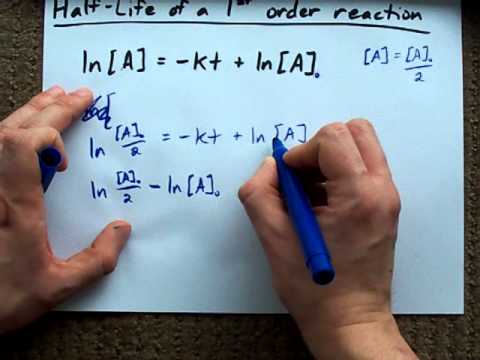
Using calculus, the differential rate law for a chemical reaction can be integrated with respect to time to give an equation that relates the amount of reactant or product present in a reaction mixture to the elapsed time of the reaction. For example, an integrated rate law is used to determine the length of time a radioactive material must be stored for its radioactivity to decay to a safe level. We can use an integrated rate law to determine the amount of reactant or product present after a period of time or to estimate the time required for a reaction to proceed to a certain extent. 
We can also determine a second form of each rate law that relates the concentrations of reactants and time. The rate laws we have seen thus far relate the rate and the concentrations of reactants. Identify the order of a reaction from concentration/time data.Define half-life and carry out related calculations.Perform integrated rate law calculations for zero-, first-, and second-order reactions.Explain the form and function of an integrated rate law. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
